
Measuring the Surface Cleanliness
Once the surface profile is at the required standard then the surface cleanliness and contamination should be addressed.
Before the surface is blast cleaned, any surface contaminants should be removed. These surface contaminants could be salts, oil, grease, cutting oils, drilling compounds and more. It would be wrong to assume that the blasting process will remove all contaminants. Whilst the contaminants may not be visible, they will have been distributed over the steel surface.
The condition of the substrate can be assessed using pictorial surface standards. These allow visible contaminants to be checked (rust). However, it is also necessary to assess the surface for non-visible contaminants, particularly water soluble salts. The presence of salts will have a detrimental effect on the life of a coating and can cause premature failure of the coating. Water jetting (the use of water under pressure) is used to remove surface contaminants on pre-coated surfaces. Water jetting will not create a profile.
When washing with water it cannot be assumed that the water is free of contamination and it is, therefore, necessary to test the water. For example, tap water will contain fluoride and/or chlorine. As mentioned earlier, the presence of chloride ions can be determined using the Elcometer 134W Test Kit. The pH value of the water can be checked using a pH meter such as the Elcometer 148 pH Tester.
The Elcometer 138 Conductivity Meter will measure the level of soluble salts in a solution and provide the user with a quantifiable measure of the salt content. Surface cleanliness can be split into five categories:
- Visible Contaminants: oil, grease, cutting oils, drilling compounds, etc.
- Dust: blast media, soil, air particulates, etc.
- Salts: all salts and in particular chloride, sulphate and nitrate ions.
- Carbamates: specifically amine blush
- pH
pH
pH is a chemical scale that relates to the concentration of free protons in a solution. Although this sounds rather complicated, the concentration of these protons give a solution its acidity, a concept which everyone is familiar with. Acidity is something that everyone can detect when tasting something, with a sharp flavour like vinegar or lemon juice. The opposite of acid, alkaline, has a soapy flavour.
The scale itself in general runs from 1 to 14. A solution with a low pH is acidic, in that it has a high concentration of protons and a solution with high pH has a particularly low concentration of protons. Pure water has a pH of 7, a state described as neutral.
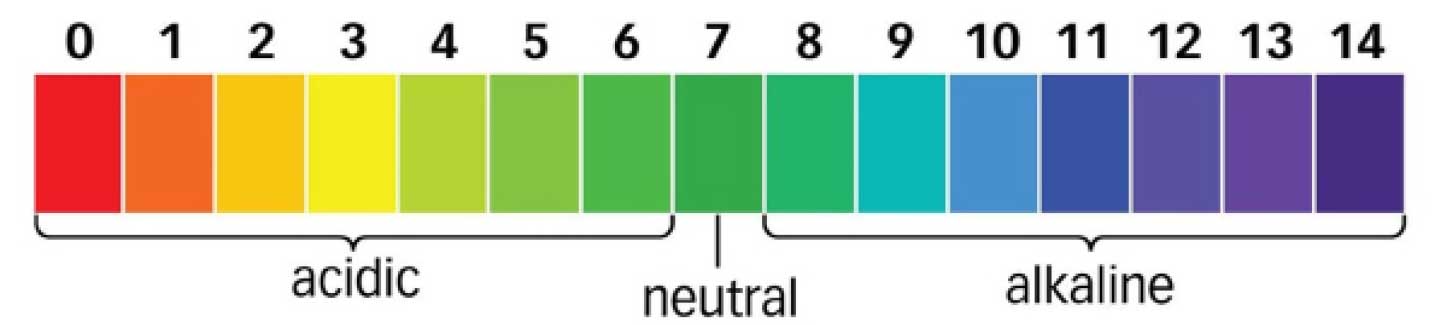
pH has a variety of effects on chemical reactions, depending on the chemical system. High acidity can catalyse or speed up some reactions and retard others depending on the exact nature of the reaction system.
In the context of corrosion of steel, a high acidity (low pH) accelerates the corrosion process. This is due to the acid increasing the rate that gaseous oxygen dissolves into the solution, where it reacts with the steel to form rust and other oxides of iron. This would be especially problematic if there are any defects in a protective coating and the pH was low when the coating was applied. This is because tiny amounts of moisture will penetrate to the surface through pinholes and will have their effect magnified by the acidic conditions, potentially creating a major corrosion problem.
Many different contaminants can affect the pH in a solution. From acid rain residue to biological deposits, they all cause a pH change on the surface. If a test were to indicate more acidic or more alkaline conditions than normal, this would show that there was some kind of contamination on the surface that may require cleaning off for optimum coating performance.
Checking for Visual Contaminants
Visual surface contaminants are, by definition, visible to the eye. In some environments, such as low light conditions, the uses of torches (flashlights) and, in particular, UV lamps (black-lights) can often identify less visible contaminants.
Measuring Dust Particles
Dust on blast cleaned surfaces will reduce coating adhesion, leading to premature coating failure and a sub-standard coating finish. A dust tape test kit, such as the Elcometer 142 Dust Test Kit, allows the assessment of both the quantity and the size of dust particles on surfaces being prepared for painting.
These kits can be used in accordance with the recommendations of BS EN ISO 8502-3, as a pass/fail test, and provide a permanent record of the presence of dust. The level of dust particles on a surface can be assessed by taking a length of sticky tape and applying it firmly to the substrate. The tape is then removed and attached to a comparison chart.

This chart will provide a visible assessment of the level of contamination. Some standards will describe a force to be used when applying the tape. Tape roller applicators are available to achieve a consistent result.
Surface Cleanliness – Salt Measurement
Before we discuss the types of measurement techniques and gauges available to measure soluble salts and ion specific salts, it is important to understand the basic principles of corrosion. Corrosion is an electrochemical process in which the metal reacts with the environment, forming an oxide. Oxidisation of a metal requires three key elements: an anode, a cathode and an electrically conducting solution (electrolyte). This is known as a corrosion cell.
The surface of the steel will naturally have numerous anodic and cathodic areas. While the surface is dry, there is no reaction between these areas and the steel will not oxidise (rust). However, moisture on the surface, which can conduct electricity and therefore act as an electrolyte, will create the corrosion cell and oxidisation will occur. Chloride ions in the liquid will increase the conductivity of the solution and therefore increase the rate of corrosion.
SSPC define soluble salts as “ionic chemical compounds that dissolve in water to form a solution of positive and negative ions”.

Soluble salts are hygroscopic – that is to say, they absorb moisture from the atmosphere. As liquid applied coatings are permeable (i.e. allows moisture to pass through), a soluble salt ion on the substrate can draw the moisture through the coating. This will create an electrolyte and a corrosion cell – allowing corrosion to commence under the coating, well before the coating visibly fails.
Furthermore, the products of a corrosion cell are acidic, adding further to the corrosion of the metal substrate. This can cause the substrate to become significantly eroded causing deep pockets (pits) in the surface to a level where the substrate itself will “fail”.
Within the coatings industry, surface contamination from salt ions such as chlorides, sulphates and nitrates have been shown to be the most detrimental – their presence leads to premature coating failures in weeks and definitely reduces the overall lifespan of the coating system. Typical coating failures observed through soluble salts include delamination and blistering.
To avoid the premature failure of a coating, it is absolutely necessary to ensure the surface (substrate or coated surface in a multi-layer system) is clean of all contaminants prior to the application of the next coating. There are two types of soluble salt measurements;
1. A test that determines the presence of all soluble salts on the surface.
2. A test that will determine which type of salt is on the surface (chloride, sulphate or nitrate).
Measuring Soluble Salts
There are a number of field test methods available for determining the level of soluble salts on a surface. All the tests available require an extraction of the salts from the surface, into a solution. The salt content is then measured in “parts per million”.
It is important to know the volume of the solution used during the extraction and the area of the tested surface. Using these two values, the level of salt can be calculated in parts per million (PPM). The PPM value can then be converted into other values such as µg/cm² (micro grammes per square centimetre) – units specified in many standards. All tests use the assumption that de-ionised water does not conduct electricity.
Consequently, the conductive properties of de-ionised water contaminated by soluble salts increases as the concentration of salts increases. The level of conductivity can be determined through the use of a conductivity meter.
The Swab Method
A pure cotton wool swab is soaked in a container filled with a defined volume (typically 10ml [0.34floz.]) of de-ionised water. The swab is then wiped over a defined area of the substrate to remove the soluble salt that may be present.
The swab is rinsed in the container of de-ionised water and the process of wiping the surface with the swab and rinsing in the de-ionised water is repeated. Typically up to 4 times.
A fresh, pure cotton wool swab is then used to dry the test area of the surface and this swab is also then rinsed in the container of de-ionised water. The repeated swabbing of the test area and rinsing in the de-ionised water is done to ensure all of the soluble salts are removed.
Finally, both swabs are left in the container for up to 2 minutes before testing the solution with a conductivity meter to determine the salt content.
The swab method is very difficult to undertake on vertical or overhead surfaces. The SSPC TU-4 standard indicates that the efficiency of retrieval of the swab test method is between 25 – 35%, making the test less sensitive to low levels of salt on the surface.
The Patch Method (Bresle Method)
The Bresle patch is a “cell” created by a latex sheet framed by a self-adhesive foam seal. The area of the cell is controlled (usually 12.5cm²/1.93 in²). The foam seal is compressible – allowing it to follow the profile of the substrate when applied – thus creating a seal on non-flat substrates. The Bresle patch is attached to the surface to be measured creating a “blister” sample area.

1.5ml of de-ionised water is injected into the patch through the foam seal using a syringe. Before removing the syringe, the needle should be repositioned and any excess air removed from the sample area, being careful to leave the water in the patch.
Then a further 1.5ml of de-ionised water is then injected into the patch. The water is then left in the patch for a pre-determined time. This will vary according to the test standard being followed:
Standard ISO 8502-6 states: The water is left for a period of time (typically up to 10 minutes) and the syringe is used to suck out the liquid and re-injected 4 times – creating a flushing action over the substrate. Finally, the liquid is extracted and tested with a conductivity meter. Note: a recent rewrite of the standard allows for the use of a latex sleeve as an alternative to the bresle patch.
Standard USN PPI 63101-000 states: The syringe is removed and the patch is rubbed for 10-15 seconds to allow the water to dissolve any contaminants. The water is then removed by re-inserting the syringe and the conductivity of the liquid is measured using a conductivity meter.

The conductivity is usually displayed in micro siemens per centimetre (μS/cm) and millisiemens per centimetre (mS/cm) and/or salinity (%). To calculate the surface density of salts as defined in the standards, it is necessary to multiply the reading by one of the following factors:
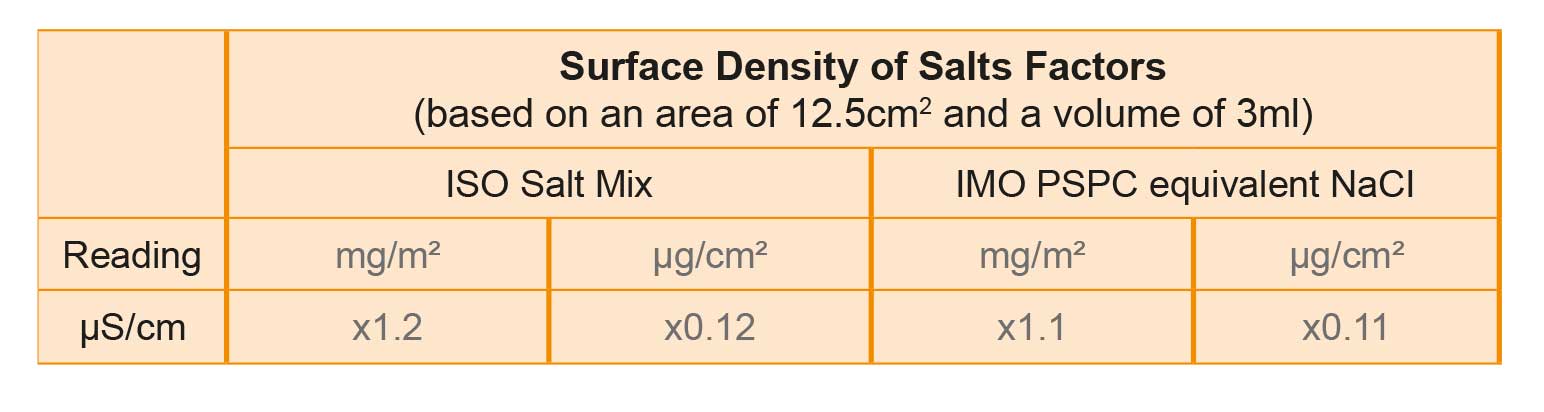
The Bresle patch method has, according to the SSPC TU-4 standard, an efficiency of retrieval of between 45 – 60%, making it more sensitive to low levels of salt than the swab test. The patch method does have some disadvantages; it requires the use of syringes, can be time consuming and presents a health and safety risk. The patch can leave an adhesive/foam residue on the substrate when removed.
Salt Contamination Meter
Two quicker and easier ways to check for contamination is to use the Elcometer 130 Salt Contamination Meter and the Elcometer 130 SSP Soluble Salt Profiler. A sterile filter paper of known area is soaked with a controlled amount of distilled water (1.6ml) and placed on the substrate to be tested, using a pair of tweezers.


After waiting for a minimum of 2 minutes, the filter paper is removed and placed in theElcometer 130 Salt Contamination Metermeter which measures the conductivity of the wet paper. The salt level is calculated and displayed in a choice of several different units including; μg/cm2, ppm, μS/cm, mS/cm, % salinity and mg/m2. A matrix of electrodes is used to assess the conductivity and hence salt content of the filter paper.
This matrix enables a more in-depth analysis of the filter paper and the contaminated water it contains. The gauge is now able to measure the conductivity between each pair of electrodes and “map” the complete area of the filter paper.

The filter paper area can be split into 4 sections, each similar in size to the area of a Bresle patch and as well as the average value for the complete filter paper, the gauge can report the values for these four sections. Independent testing has been carried out that shows that these readings are in fact equivalent to Bresle patch results.
Salt does not evenly distribute itself across a surface. There will always naturally be areas of higher or lower concentrations of salt and the Elcometer 130 SSP Soluble Salt Profiler allows the user to see how the salt is distributed across the test area.
The ability to view the salt density map informs the user that although the test piece may achieve a level of contamination that is low enough to satisfy the specification, there may be areas that are more prone to local corrosion in the future.
These areas can be highlighted further by using the pass/fail mode. The user can apply a limit to the gauge and the salt levels are then displayed as red or white areas indicating a pass or a fail together with the highest and lowest values measured.
Soluble Salt Meters
There are several soluble salt meters on the market that ‘automate’ the Bresle patch method in one form or another. The SSM-21P, for example, is used extensively in the Japanese marine sector as an alternative to Bresle.
A chamber/cell is attached to the substrate using internal magnets. The chamber is filled with 3ml of de-ionised water via a syringe or dosing unit. A propeller or plunger then agitates the liquid over the substrate, washing the surface.
Once the agitation process has been completed, internal sensors within the chamber measure the conductivity and temperature of the liquid and the gauge displays the conductivity level.
A benefit of the Elcometer 130 Salt Contamination Meter over other soluble salt test methods is that the test method does not require the inspector to be present for the duration of each individual test. This allows the inspector to set up other salt tests and come back later to the previous tests to undertake the measurement; speeding up on-site measurement.
Considering that some test methods/standards require a defined number of tests to be undertaken per area, this can result in a dramatic saving of time. USN PPI 63101-000, for example, states that the number of Bresle tests taken should be as follows:
- Five (5) measurements every 90m² (1000ft²)
- Areas less than 90m² (1000ft²) shall have five measurements made
Measuring Ion Specific Salts
The environment/atmosphere we live in is filled with naturally occurring and man-made salts. It has been established that the key salts that are the most detrimental to coatings are chlorides, sulphates and nitrates. Other industries may, of course, have concerns over others.
Structures can be exposed to chlorides from marine environments (the sea - sea salt is sodium chloride), water treatment (chlorinated water) and, surprisingly, desert environments can also be chloride rich as many were former seas.
Sulphates, from burning fossil fuels, rich in sulphur (gas, diesel, coal etc.) and nitrates from fertilisers and automotive engines are all converted into weak sulphuric and nitric acid respectively. This creating acid rain when coming into contact with the moisture in the air – all corrosive on contact with metal substrates. There are several ion specific tests which are available as either specific ion tests or combined into ion test kits.
Chloride Ion Test
In order to test the surface for chloride contamination, there are two possibilities. TheElcometer 134S is a chloride ion specific test kit for measuring the level of chloride on surfaces. It works in a similar way to the Elcometer 134 Abrasive and Water Tests, discussed earlier in the previous article.
A latex sleeve with a self-adhesive foam sealing ring is filled with an extract solution and adhered to the surface being tested. The solution is manipulated against the surface to dissolve and extract the salts. Once the sleeve is removed, a titration tube is then inserted and the liquid is assessed as before.
Chloride test strips are also available which can be used to measure the chloride content of the de-ionised/soluble salt solutions from the methods discussed above. A strip is placed into the solution and the fluid rises up the strip until completely saturated. The strip contains silver ions which combine with chloride in the sample to form a white column of chloride. The concentration level can then be determined using the printed scale on the strip.

Fig 11. The Elcometer 134S latex sleeve

Chloride, Sulphate and Nitrate Ion Test Kit
The Elcometer 134 CSN Test Kit offers a single kit solution for the field testing of chloride, sulphate and nitrate specific ions. Using pre-measured chemical tests, the surface salts are extracted using the sheath method discussed previously. Chloride ions are then measured using the Elcometer 134 method. Sulphates are measured using a colourimeter and nitrates are measured using a nitrate specific test strip (supplied with the kit).

Measuring other forms of Surface Contamination
As has been discussed above, sulphates and nitrates, when mixed with the moisture in the environment, create weak sulphuric and nitric acids – corrosive to structural metals. pH meters and pH test strips, such as the Elcometer 148 pH Tester and the Elcometer 138 - PH can be used to determine the acidity of a liquid.
Measuring Amine Blush
Amines and amine functional amides are one of the most common curing agents for high performance epoxy coatings. In coating applications, amine cured epoxy formations generally can be cured at room temperature to yield films with high mechanical strength; good water, chemical and corrosion resistance, as well as excellent adhesion properties.
The problem of amine blush can occur when using amine cured epoxy resin coatings in a multi-layer system if the original coating cures in a low ambient temperature and/or in a high humidity environment. Amines that have migrated to the coating surface can react with the carbon dioxide (CO2) present in the air, ultimately resulting in the formation of a film of carbamates (amine blush) on the surface of the epoxy coating.
The presence of carbamates will lead to inter-coat adhesion failures if the film is not removed before re-coating. It is therefore important to determine if the coating exhibits the phenomenon of amine blush when:
1. Determining if an amine cured epoxy coating can be over coated
2. Analysing why an epoxy coating could not be successfully over coated
Amine blush can sometimes (but not often) be visible to the naked eye - appearing as a thin spotty or greasy layer ranging in colour, from white to amber. The Elcometer 139/A Amine Blush Swab and Elcometer 139/C Amine Blush Chip Screen Test Kits provide the user with a clear indication of whether amine blush is present.
The Amine Blush Swab Test Kit provides users with a field based test, whilst the Amine Blush Chip Screen Test Kit allows coating samples to be removed and tested in an office or sent to a laboratory for further analysis.
After the surfaces are tested for contamination and are found to be within specification, then providing the climatic conditions are within the specified limits for coating application, then the surface can be coated.



